Equiptrack products and services are based only on information supplied to Equiptrack. Equiptrack does not have the complete history of every unique piece of equipment. Use the Equiptrack search as one important tool, along with an equipment inspection and functional check, to make a better decision about purchasing your next piece of used medical equipment.
Medical Device Recall: BD PosiFlush Prefilled Saline Syringe (10mL Syringe with 10mL Saline Fill)
Medical Device Recall: BD PosiFlush Prefilled Saline Syringe (10mL Syringe with 10mL Saline Fill)
Introduction:
BD has issued a Medical Device Recall for an incorrect stopper which was used in nine lots of 10mL BD PosiFlush Normal Saline Flush Syringes with General Pump Compatibility. The use of BD PosiFlush Prefilled Flush Syringes with this stopper causes an increase in injection force, which has the potential to trigger alarms if used with a syringe pump.
FDA Recall Links:
Recall Scope:
Lot Numbers: 3207548 3207555 3214826 3226386 3214828 3226388 3250302 3250303 3250304
Introduction:
BD has issued a Medical Device Recall for an incorrect stopper which was used in nine lots of 10mL BD PosiFlush Normal Saline Flush Syringes with General Pump Compatibility. The use of BD PosiFlush Prefilled Flush Syringes with this stopper causes an increase in injection force, which has the potential to trigger alarms if used with a syringe pump.
FDA Recall Links:
Recall Scope:
Lot Numbers: 3207548 3207555 3214826 3226386 3214828 3226388 3250302 3250303 3250304
Reason for Recall:
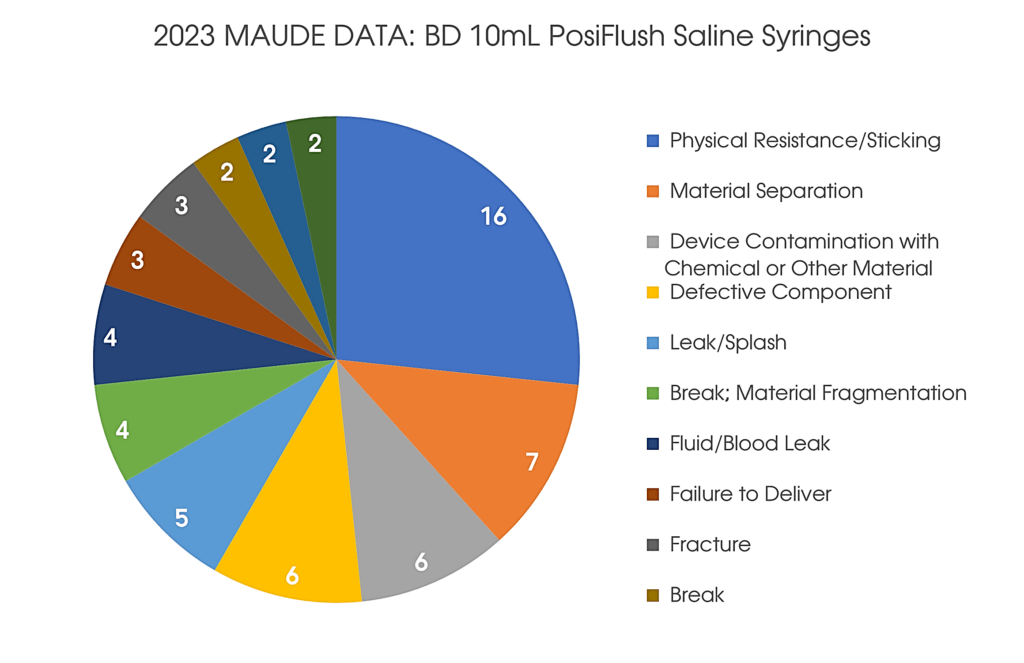
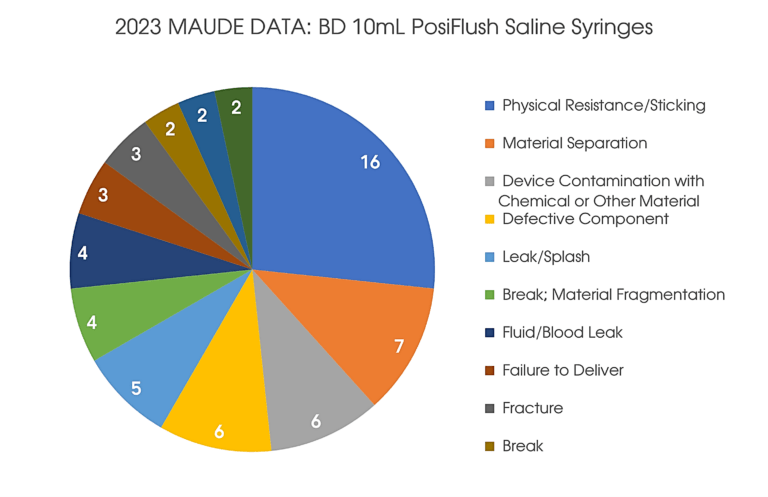
An incorrect stopper was used in nine lots of 10mL BD PosiFlush Normal Saline Flush Syringes with General Pump Compatibility. The use of BD PosiFlush Prefilled Flush Syringes with this stopper causes an increase in injection force, which has the potential to trigger alarms if used with a syringe pump.
FDA’s Determination:
This Recall is under investigation by the firm.
Recall Quantity:
5,085,120 units
Customer/Patient Actions:
1. Immediately review your inventory for the affected product. a. If the product is used for syringe pumps, destroy all affected product subject to this field action following your institution’s process for destruction. b. If the product is used for manual application, the product can continue to be used per IFU.
2. If the affected product was previously used on a patient, no further actions are needed.
3. If the affected product is currently in use with a syringe pump, replace the syringe with a non-impacted lot. Please note that healthcare facilities and providers should rely on their syringe pump manufacturer’s IFU for the specific syringes that are compatible with their syringe pumps.
4. Share this field action notification with all users of the product within your facility network to ensure they are also aware of this field action.
5. Complete the attached Customer Response Form and return to the BD contact noted on the form whether or not you have any of the impacted material so that BD may acknowledge your receipt of this notification per FDA requirements and subsequently process your product replacement if product is used on syringe pumps and has been discarded.
6. Report any adverse health consequences experienced with the use of this product to BD. Events may also be reported to the FDA’s MedWatch Adverse Event Reporting program via: Web: MedWatch website at www.fda.gov/medwatch Phone: 1-800-FDA 1088 (1-800-332-1088) Mail: MedWatch, HF-2, FDA, 5600 Fishers Lane, Rockville, MD 20852-9787
Distributor Actions:
1. Immediately review your inventory for the affected product. Destroy all unused product subject to the product advisory following your institution’s process for destruction.
2. Identify all customers within your distribution network that purchased any affected product as defined in this notification.
Contact Information:
If you have any questions or concerns regarding this notification, please contact BD or your local Service Representative.


